- Description
- Who gets Giant Cell Arteritis (the “typical” patients)?
- Classic symptoms of Giant Cell Arteritis
- What causes Giant Cell Arteritis?
- How is Giant Cell Arteritis diagnosed?
- Treatment and Course of Giant Cell Arteritis
- What’s new in Giant Cell Arteritis?
- In medical terms, by David Hellmann, M.D.
Description
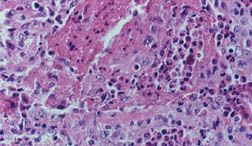
Giant cell arteritis (GCA) is the most common form of vasculitis that occurs in adults. Almost all patients who develop giant cell arteritis are over the age of 50. GCA commonly causes headaches, joint pain, facial pain, fever, and difficulties with vision, and sometimes permanent visual loss in one or both eyes. Because the disease is relatively uncommon and because the disease can cause so many different symptoms, the diagnosis of GCA can be difficult to make. With appropriate therapy, GCA is an eminently treatable, controllable, and often curable disease. The disease used to be called “temporal arteritis” because the temporal arteries, which course along the sides of the head just in front of the ears (to the temples) can become inflamed. However, we also know that other blood vessels, namely the aorta and its branches, can also become inflammed. The term “giant cell arteritis” is often used because when one looks at biopsies of inflamed temporal arteries under a microscope, one often sees large or “giant” cells.
Who gets Giant Cell Arteritis?
GCA is a disease of older people. The average age at onset is 72, and almost all people with the disease are over the age of 50. Women are afflicted with the disease 2 to 3 times more commonly than men. The disease can occur in every racial group but is most common in people of Scandinavian descent.
Classic Symptoms of Giant Cell Arteritis
The most common symptoms of GCA are headache, pain in the shoulders and hips (called polymyalgia rheumatica), pain in the jaw after chewing (called jaw claudication), fever, and blurred vision. Other symptoms can include tenderness of scalp (it hurts to comb the hair), cough, throat pain, tongue pain, weight loss, depression, stroke, or pain in the arms during exercise. Some patients have many of these symptoms; others have only a few. Blindness — the most feared complication — can develop if the disease is not treated in a timely fashion.
What Causes Giant Cell Arteritis?
We do not know. We do know that aging has something to do with the disease. And we know that the body’s immune system attacks and inflames the arteries. But we do not know why the immune system attack occurs when and where it does.
How is Giant Cell Arteritis Diagnosed?
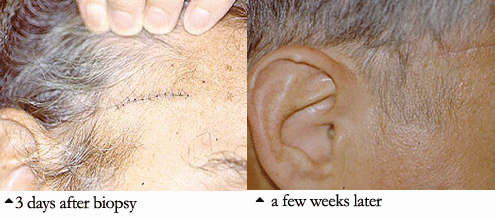
The diagnosis is made by doing a biopsy of the temporal artery. Using a local numbing medication (the same one used by a dentist), the doctor can remove a small part of the temporal artery from under the scalp and look at it under the microscope for evidence of inflammation. A temporal artery biopsy is almost always safe, causes very little pain, and often leaves little or no scar. An example of this is pictured below
There are blood tests that help the doctor decide who is likely to have GCA. Almost everyone with the condition has an elevated erythrocyte sedimentation rate (also called “sed rate”). The sed rate measures how fast a patient’s red blood cells settle when placed in a small tube. In inflammatory conditions, red blood cells settle more quickly than in non–inflammatory states. In addition, most patients with GCA have a slight–anemia, or low red blood cell count. Other conditions can also cause a high sed rate or anemia, so the final diagnosis depends on a temporal artery biopsy.
A few patients with GCA do not have positive biopsies. We now know that GCA does not affect every part of every temporal artery but can “skip” around. When one biopsy is negative, biopsying the temporal artery on the other side can lead to the diagnosis.
Treatment and Course of Giant Cell Arteritis
GCA requires treatment with prednisone, a type of corticosteroid. Typically, treatment begins with 40–60 mg of prednisone, taken by mouth each day. Most patients improve rapidly and dramatically on this dose, with improvement of most symptoms in 1–3 days. Unfortunately, if blindness has occurred as a symptom it is usually irreversible, which only emphasizes the importance of early detection and treatment.
Almost all patients experience side effects from prednisone. After the patient improves, the doctor gradually reduces the prednisone dose. The rate of tapering prednisone depends on how the patient feels, what the doctor finds on exam, and the results of blood tests, including the sedimentation rate. Although virtually all patients are able to reduce their prednisone dose, most require some amount of prednisone for 1–2 years. Longer treatment periods are not uncommon.
In medical terms, by David Hellmann, M.D.
A discussion of Giant Cell Arteritis written in medical terms by David Hellmann, M.D. (F.A.C.P.), Co-Director of the Johns Hopkins Vasculitis Center, for the Rheumatology Section of the Medical Knowledge Self-Assessment Program published and copyrighted by the American College of Physicians (Edition 11, 1998). The American College of Physicians has given us permission to make this information available to patients contacting our Website.
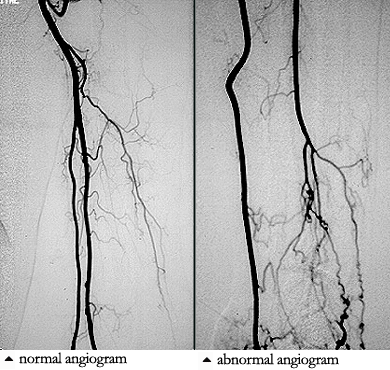
Giant cell arteritis is vasculitis of unknown cause that affects the elderly and is characterized by panarteritis of medium- to large-sized arteries, especially in the extracranial branches of the carotid artery. The average age of onset is 72 years, and women are affected two times as often as men. Irreversible blindness, the most commonly feared complication, results from necrosis of the posterior ciliary branch of the ophthalmic artery and is usually preventable by early diagnosis and corticosteroid treatment.
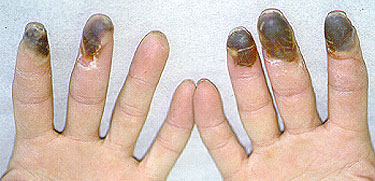
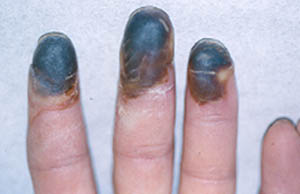
Giant cell arteritis can begin suddenly or gradually with nonspecific symptoms such as malaise, weight loss, depression, and fatigue or with the classic symptoms of headache, scalp tenderness, jaw claudication, visual changes, or polymyalgia rheumatica. Polymyalgia rheumatica which can occur with or without giant cell arteritis, is characterized by pain and stiffness of the hips and shoulders that worsens in the morning. About one third of patients resemble the preceding patient and present with atypical manifestations such as fever of unknown origin, respiratory symptoms (dry cough is most common), large vessel disease (causing Raynaud’s phenomenon, claudication, or thoracic aortic aneurysm), mononeuritis mutiplex, glossitis, or profound anemia. Although giant cell arteritis accounts for only 2% of all fever of unknown origin, it accounts for 16% of fever of unknown origin in patients over age 65 years and is often associated with rigors and sweats. Only half of patients have enlarged, nodular, or nonpulsatile temporal arteries: normal temporal arteries on physical examination do not exclude the diagnosis. Subclavian bruits, diminished pulses, aoritic regurgitation, or Raynaud’s phenomenon are found in patients with large vessel disease. Fundoscopic examination is normal in the first day or two after blindness develops. Almost all patients have a markedly elevated ESR, averaging about 100 mm/h. Very rarely, the ESR may be normal, especially in patients who are already taking prednisone for allergic or respiratory diseases. MOst patients have mild normochromic normocytic anemia, and 20% to 30% resemble the preceding patient in having mildly elevated serum alkaline phosphatase. The leukocyte count at presentation is usually normal, a point favoring giant cell arteritis over infection or malignancy.
Because blindness from giant cell arteritis is almost irreversible, treatment with 40 to 60 mg of prednisone should be started as soon as the diagnosis is suspected. Although immediate temporal artery biopsy has been preferred, one study suggests that biopsy remains positive within at least the first 2 weeks of corticosteroid therapy. Therapy should not be held pending biopsy. In patients with giant cell arteritis, arterial involvement is patchy: therfore, maximizing the chance of diagnosis requires obtaining a long (3 to 4 cm) segment and examining multiple sections. Positive biopsy specimens show infiltration of the vessel wall with mononuclear inflammatory cells and giant cells, intimal proliferation, and thrombosis. Unilateral biopsy specimens are positive in approximately 85% of patients, and bilateral biopsy specimens are positive in 95%. Patients dramatically improve within 24 to 72 hours of beginning therapy, and the ESR usually normalizes within 1 month. Thereafter, prednisone can be tapered slowly, although most patients require some prednisone for at least 9 months and often longer.
Treatment decisions should probably be based on the patient’s symptoms, the hemoglobin, the ESR: ESR alone should not dictate therapy. Because compression fractures develop in one third of patients, prevention and treatment of osteoporosis should be part of initail management. Methotrexate, azathioprine, and cyclophosphamide have been used in rare patients who do not respond to adequate prednisone. Long–term follow–up is required to detect late recurrences (including the late onset of thoracic aortic aneurysms with aortic regurgitation, congestive heart failure, and aortic dissection). Patients with polymyalgia rheumatica but no symptoms of giant cell arteritis above the neck (such as jaw claudication, headache and visual symptoms) do not need temporal artery biopsy and respond to low–dose prednisone (10 to 20 mg/d orally). Because polymyalgia rheumatica is a clinical diagnosis, other conditions such as hypothyroidism, amyloidosis, rheumatoid arthritis, and malignancy should be considered in the initial evaluation and reconsidered if the patient does not improve rapidly on prednisone.