What is colchicine?
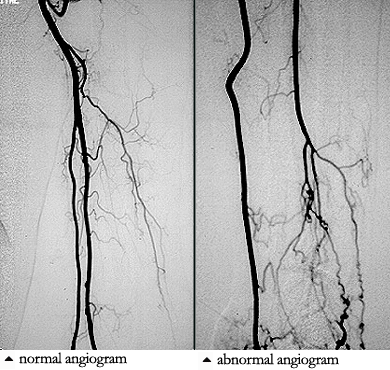
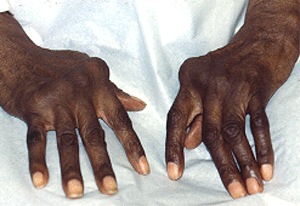
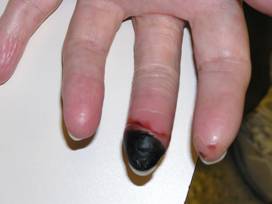
Colchicine is an oral drug used in the treatment of some forms of cutaneous vasculitis. It is a very old medicine that is more frequently encountered in the treatment of gout.
How does colchicine work?
Colchicine seems to work by preventing immune cells from becoming fully activated.
How is colchicine given?
Colchicine is given as an oral pill at a dose of 0.6 mg either once or twice per day.
Side effects:
In contrast to most other vasculitis treatments, colchicine is not an immunosuppressant drug and does not cause any significant risk of infection.
Colchicine can cause gastrointestinal side effects and requires monitoring during its use. Some patients with kidney disease may not be able to safely take colchicine on a long-term basis.